Now, mask factories in European and American countries are facing some pressure: on the one…
Medical Face Mask Making Machine: The Complete Guide for the Medical Masks Production
With the new COVID going on, mask factories are gradually established in various places around the world, so as important surgical mask making machines for mask factories, how does the surgical mask making machine produce masks? Today, TESTEX will share with you.
1. The structure and functions of disposable medical surgical masks
Common medical surgical masks are mainly composed of three layers of non-woven fabric. The inner layer is a common non-woven fabric, which is mainly used to absorb the moisture and moisture released by the wearer; the outer layer is a waterproof non-woven fabric, which is mainly used to isolate the liquid sprayed by the patient; the filter layer in the middle is used for the polypropylene melt-blown non-woven fabric treated with electret serves as a barrier against germs.
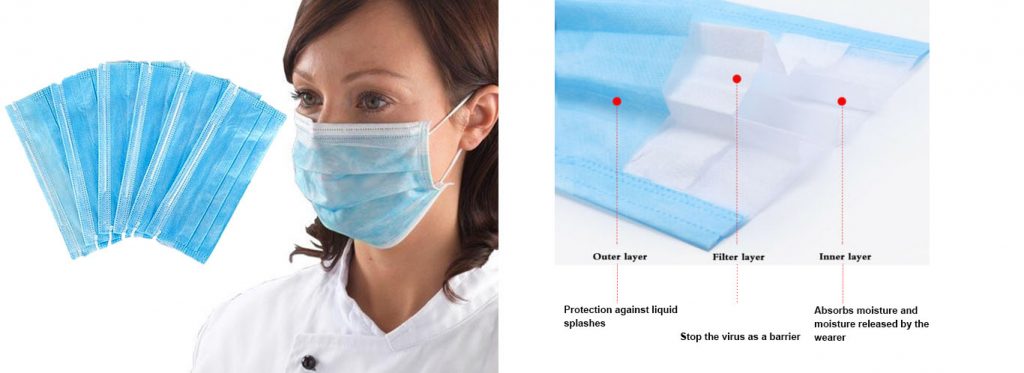 The core raw material of medical masks is polypropylene melt blown non-woven fabric after electret treatment *. It is important to remember these two keywords, as this is the key to filtering the new crown virus aerosol.
The core raw material of medical masks is polypropylene melt blown non-woven fabric after electret treatment *. It is important to remember these two keywords, as this is the key to filtering the new crown virus aerosol.
2. Mask effectiveness test and standard requirements
Common testing standards for masks are as follows (EU, USA, China)
• EN 149 “Respiratory protective devices – Filtering half masks to protect against particles – Requirements testing marking”
• EN 14683 “Medical face masks – Requirements and test methods”
• ASTM F2100 “Standard Specification for Performance of Materials Used in Medical Face Masks”
• CFR 42 Part 84 “NIOSH Guide to the Selection and Use of Particulate Respirators”
• EN 14168 “Medical face masks. Requirements and test methods”
• GB 2626 “Respiratory protective equipment. Non-powered air-purifying particle respirator”
3. The production process of surgical mask making machine
Hang the non-woven raw materials on the mask sheeter rack of the surgical mask making machine, and the surgical mask-making machine will produce it automatically after debugging. The mask body will come out, and then the mask body will be transferred to the ear band machine for spot belting. After production, it is sterilized by ethylene oxide and left for 7 days to volatilize. The specific process is as follows:
1) Combine three layers of non-woven fabric
Three different non-woven fabrics were placed on the fixed support on the surgical mask making machine and neatly stacked together by the laminator above. There is a trumpet-shaped feeding port here, and a string of wires is continuously passed through the trumpet to the laminating machine.

Adult masks have uniform size specifications. How does it ensure that different people have the right size requirements to ensure that the nose, mouth, and chin are wrapped to the greatest extent? This is due to the design of the surgical mask making machine shown below:

On the surgical mask-making machine, there is a folding device to achieve this purpose.
2) Stitch the metal wire fixed by the nose clip into the laminated three-layer non-woven fabric
From the suggestions for wearing masks correctly, we can know that the nose clip must be pinched to the bridge of the nose when wearing so that wearing the mask will be firm. Otherwise, without this structure, my nose leak is more serious, the seal is not tight, and the protective effect is affected …
From the figure below, we can see that the nose wire is matched and conveyed along one side of the non-woven fabric, then the next edge is rolled, the back is stitched, and the nose wire is stitched in.

3) Mask body unit forming
Next, in order to create a crease and better processing in subsequent processes, it needs to be flattened by a roller machine and then cut to a single mask unit.

After getting a single mask body with a reinforced edge, the next step comes.

4) Fix the hanging ear rope to the four corners of the mask with the thermoforming device
Because the process of wearing a mask, the hanging ear rope is in a relatively tight state for a long time. In order to strengthen the binding strength of the rope and the mask body, it is necessary to use adhesives at the four corners of the mask. During the pressing process, it is mechanically transported. Hang the ear rope, and fix the ear rope to the mask with an adhesive under the heat pressing device.
Through the above steps, even if a flat disposable nonwoven mask is produced, it cannot be called a disposable medical nonwoven mask here, because there is still a very important link that has not been completed. Press :

5) Earloop folding
Surgical mask-making machine automation generally refers to the automation of face mask production, if you want to save further labor you also need to connect a mask packaging machine and achieve full automation, and the key here is the folding of the earloop. Advanced mask-making machine manufacturers will integrate mask production and mask packaging to greatly improve the efficiency of the mask production line.

6) Disinfection
During the entire production process of the mask, not only the processing contact of the mask making machine but also the contact of many artificial links, the mask will inevitably be contaminated by bacteria. If it is an ordinary mask, it does not need to be sterilized, but the medical treatment requires the manufacturer to use a ring. Oxygen ethane (EO) sterilizer is disinfected. The mask was placed in an environment of 400 mg / L of ethylene oxide, and alkylation was applied to the hydroxyl group to make the microbial macromolecules inactive to achieve the purpose of sterilization. However, ethylene oxide is not only flammable and explosive, but also toxic to the human body. Therefore, it needs to stand for 7 days for analysis after disinfection. After the EO residual amount is lower than the required value, it can be packaged and shipped to medical staff.
Only medical masks produced according to the above process use high-melt melt-blown non-woven fabrics to produce filter materials, perform electret treatment to increase electrostatic adsorption, disinfect with ethylene oxide after production, and stand for 7 days to analyze ethylene oxide. Is a qualified, safe medical mask that can be used to isolate the transmission of new crown virus.
4. Required the surgical mask making machine for production
Medical Face Mask Making Machine:

For mask factories that produce disposable medical masks, the surgical mask making machine needed includes mask forming machines, mask crimping machines, mask trimming machines, nose bridge line fitting machines, and earloop welding machines.
Note: The mask production line equipment can only produce disposable flat masks, and cannot produce masks with three-dimensional or N95.
5. How to control the product quality and safety of disposable masks
1) In view of the fact that the quality of filter materials for medical masks is difficult to control through convenient and effective inspection methods, the standardized operation of the production quality management system is the main means for enterprises to ensure the quality of mask products, so they are audited in the registration technology review and system assessment. Inspectors and inspectors will pay attention to the production process and source of supply of filter materials. Enterprises should control the filtering materials of products, clarify the sources and quality requirements of filtering materials, and have relatively stable production processes and sources of supply to ensure the quality of the products. Here, some reliable testing equipment is recommended for incoming inspection. Raw materials: Need to carry out sampling inspection on the raw non-woven fabric. The sampling process will use a circular sample cutter and fabric scale to determine the weight of the non-woven fabric, also measuring the waterproof(spray rating tester) and breathable performance of the outer layer of the mask, and the moisture absorption of the inner layer. Breathability to determine whether the quality of the cloth can meet the requirements. And the packaging link: In determining whether the number of boxes is accurate, the factory generally checks by simple weighing.
2) The safety and effectiveness of medical masks are just some of the factors that play a protective role. The correct use and wearing method of the mask also directly affect the protection effect. Therefore, all the information required by the user should be clearly stated in the instructions to avoid misuse of the mask and reduce the risk of cross infection. For example, indicate the wearing method, clearly identify the front and back of the mask, suggest the use time, indicate the filter material level or related instructions, and so on. In addition, medical personnel have not yet clearly understood the scope of application of different types of medical masks. The scope of application of masks should be clearly stated in the instructions and the training of medical personnel should be strengthened.
3) Sterile medical masks are produced under the condition of a class 100,000 clean factory building, which is characterized by a maximum allowable number of dust particles equal to or greater than 0.5 microns and the number of particles must not exceed 3.5 million, and the number of particles greater than or equal to 5 microns cannot exceed 20,000. In addition, the maximum allowable number of microorganisms, the number of planktonic bacteria should not exceed 500 per cubic meter; the number of sedimentary bacteria should not exceed 10 per culture dish. At the same time, the pressure difference between clean rooms of the same cleanliness level is the same. For different cleanliness, the pressure difference between adjacent clean rooms is ≥5Pa, and between clean rooms and non-clean rooms is ≥10Pa. This is mainly to ensure that the air flows from the clean area to the non-clean area, and to avoid the backflow of airflow. The temperature is generally controlled at 20-22 ° C in winter; 24-26 ° C in summer; fluctuation ± 2 ° C. The humidity of clean room in winter is controlled at 30-50%, and the humidity of clean room in summer is controlled at 50-70%. When there are no special requirements for temperature and humidity, it is advisable to wear clean work clothes without producing a comfortable feeling. For its functional layout, equipment and management requirements, please refer to the “Guide to Inspection Points for Clean Room (District) of Medical Devices”.
If you think this post is helpful, please help to share it.
This Post Has 26 Comments
Leave a Reply
You must be logged in to post a comment.








I want to set up a mask manufacturing unit( surgical,ffp1 and n 95).pls guide us how much space needed and which machines needed and raw material needed.
Pls shar any business plan u have .i m from chandigarh,india
Thank you for your message, we will reply you asap!Best regards.
please send price ,particulars contact 9344680097
Thank you for your message, we will reply you asap!Best regards.
Please send price of semi & Auto mask making machines
Thank you for your message, could you please leave your country, then we’ll contact you asap!Best regards.
Do you ship the machinery to USA? Do you also supply raw materials to make surgical masks level 1, 2 and 3. We want to set up manufacturing unit in America.
Hello dear, our sales colleague will contact you soon, kindly wait please, thanks and regards!
I went surgical mask machine
Hi Pankaj garg, Thank you for the inquiry, our sales will contact you soon.
I plan to set up a face mask manufacturing division. I like to learn how to go about it.
Hi Keith, You are at the right place at the right time, we have a full guide on how to set up a face mask manufacturing factory, our sales engineer will contact you soon. Thank you!
[…] Did you know? Without electrostatic treatment, most materials that have 1um or better filtration compromise breathability. This is why the materials with the best particle filtration are often ill-suited for use in DIY masks. The special melt-blown filter materials that are used in N95 masks to get great filtering require special treatments (e.g. Corona Treatment or Electret Treatment) to achieve optimal particle filtration performance. More information on medical mask production. […]
Thank you , very useful information. Please advise your current delivery time and forward by mail standard applicable to mask production. You have your machine working in India near New Delhi, in that case advise if we observe the machine in working.
Hi Suraj,
Our sales engineer might have contacted you already, if not, please call us at +86 769 2329 4842, thank you.
Hi,
We wish to ask about the gsm needed for the production of Type I, Type II and Type IIR according to CE. Any other process/procedure is needed in order to comply with the above type rating.
Thanks.
Best regards,
Che
Hi Choong, our sales engeer might have alreay talked to you abou the answer, if not, please contact us via email: [email protected], or call us at +86 769 2329 4842, thank you for your comment.
Hi Che, thank you for your comment, our sales engineer should have contacted you already, if not, please try to contact us via email: [email protected], or call us +86 769 2329 4842, thanks.
For purchase automatically mask making machine
Please quote your price for Tm120-medical-mask-production-line including cost of ianding.
Hi, pls send mask machine product catalogue/brochures.
Product specifications with quotation.
I would like to know further information on disposable medical mask production in Australia.
Did you give the drawing to be manufactured in Turkey olsunn mask machine possible. If your answer is yes, what is the price
how to make 3 different layers of facemask
The semi automatic machine costing how much?.
How may days will takes for manufacturing this machine?
I am from Malaysia.. interested in your semi automatic machine.
Dear Sir,
My company is interested to serve the nation and therefore want to first start by manufacturing protective face masks for public use to prevent from dangerous viruses like COVID-19.
Please guide me on how to go ahead and start the manufacturing process and any business plan you can share o my email ID.
Thank you & Regards,
Raveen Bhatnagar
Cell:+91-7738034190